
Scheme 1 | Catalytic activity of a supported palladium–benzimidazole complex toward alkene hydrogenation | SpringerLink

PDF) An Efficient Synthesis of Benzimidazoles via Palladium-Catalyzed Amine Exchange Reaction from Trialkylamines to o-Phenylenediamine in an Aqueous Medium
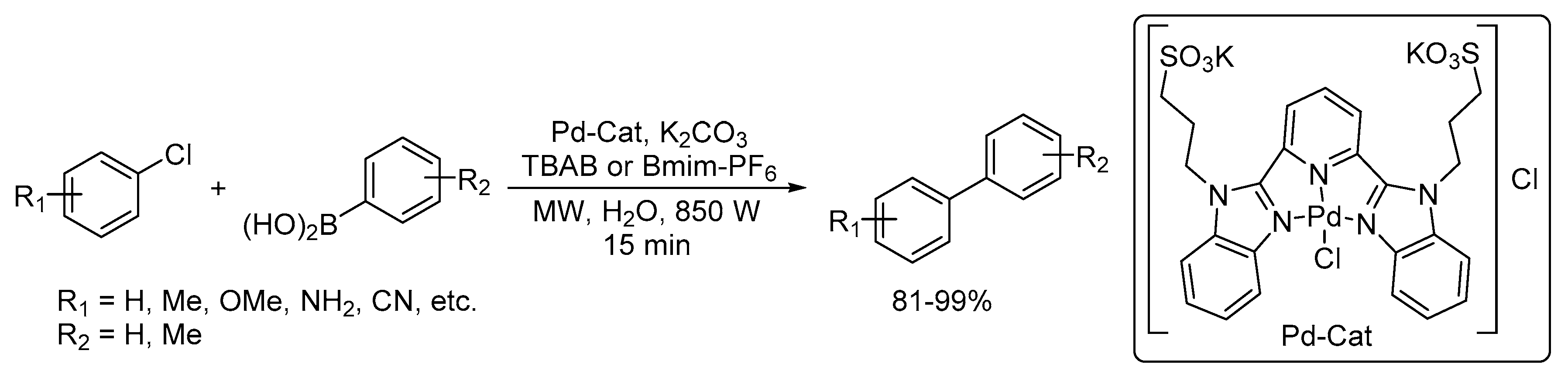
Catalysts | Free Full-Text | Microwave-Assisted Palladium-Catalyzed Cross-Coupling Reactions: Generation of Carbon–Carbon Bond | HTML

Synthesis, crystal structures and biological activities of palladium(II) complexes of benzimidazole and 2-methylbenzimidazole - ScienceDirect

Arylation of heterocyclic compounds by benzimidazole-based N-heterocyclic carbene-palladium(II) complexes - ScienceDirect
![1-Methyl-3-[1-[2-(3-methylbenzimidazol-1-yl)naphthalen-1-yl]naphthalen-2-yl] benzimidazole;palladium(2+);2,2,2-trifluoroacetate | C40H26F6N4O4Pd - PubChem 1-Methyl-3-[1-[2-(3-methylbenzimidazol-1-yl)naphthalen-1-yl]naphthalen-2-yl] benzimidazole;palladium(2+);2,2,2-trifluoroacetate | C40H26F6N4O4Pd - PubChem](https://pubchem.ncbi.nlm.nih.gov/image/imgsrv.fcgi?cid=24874065&t=l)
1-Methyl-3-[1-[2-(3-methylbenzimidazol-1-yl)naphthalen-1-yl]naphthalen-2-yl] benzimidazole;palladium(2+);2,2,2-trifluoroacetate | C40H26F6N4O4Pd - PubChem
![PDF] catena-Poly[[bis(1H-benzimidazole-κN 3)palladium(II)]-μ-benzene-1,4-dicarboxylato-κ2 O 1:O 4] | Semantic Scholar PDF] catena-Poly[[bis(1H-benzimidazole-κN 3)palladium(II)]-μ-benzene-1,4-dicarboxylato-κ2 O 1:O 4] | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/08fac28127f1e880caa63d6e1acc0cab376947a4/1-Table2-1.png)
PDF] catena-Poly[[bis(1H-benzimidazole-κN 3)palladium(II)]-μ-benzene-1,4-dicarboxylato-κ2 O 1:O 4] | Semantic Scholar

Formation of polynuclear palladium complexes with the benzimidazole-2-thiolate anion | Semantic Scholar

PDF) 5-Nitrobenzimidazole containing Pd(II) catalyzed C-C cross-coupling reactions: The effect of the N-substituent of the benzimidazole structure on catalyst activity
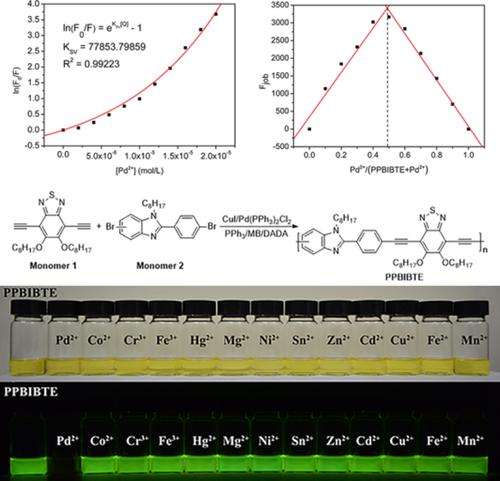
A Novel Conjugated Polymer Consists of Benzimidazole and Benzothiadiazole: Synthesis, Photophysics Properties, and Sensing Properties for Pd2+ - J. Polym. Sci. A Polym. Chem. - X-MOL
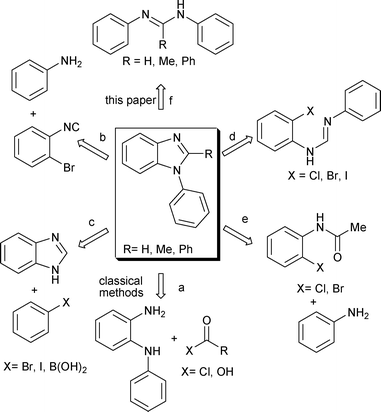
Palladium -catalyzed aerobic oxidative C–H amination: synthesis of 2-unsubstituted and 2-substituted N -aryl benzimidazoles - RSC Advances (RSC Publishing) DOI:10.1039/C2RA20328F
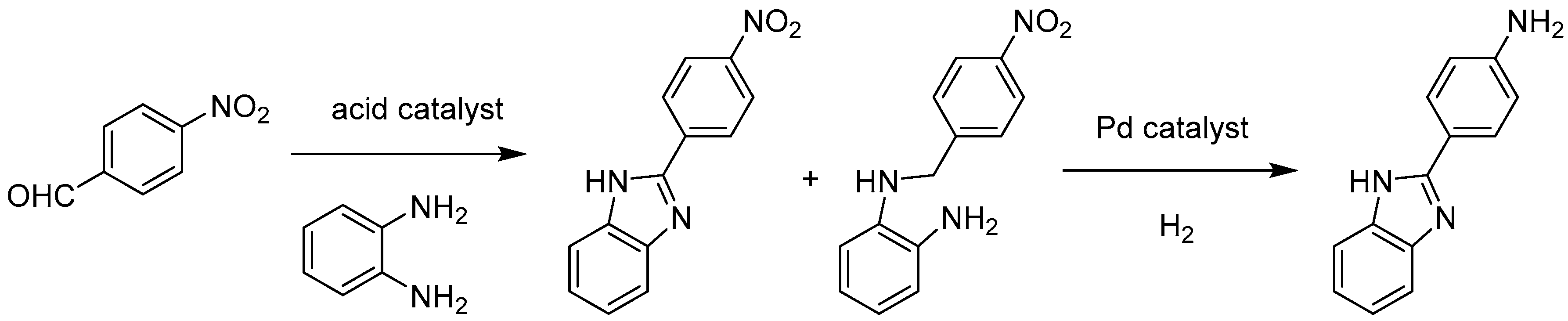
Molecules | Free Full-Text | Encapsulation of Metal Nanoparticles within Metal–Organic Frameworks for the Reduction of Nitro Compounds | HTML
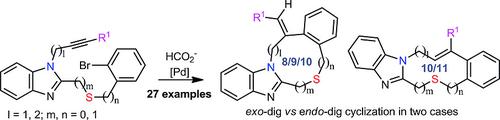
Synthesis of Benzimidazole‐Fused Medium‐Sized N,S‐Heterocycles via Palladium‐Catalyzed Cyclizations,European Journal of Organic Chemistry - X-MOL

PDF) Heck reaction using palladium-benzimidazole catalyst: synthesis, characterisation and catalytic activity

Carbohydrate triazole tethered 2-pyridyl-benzimidazole ligands: Synthesis of their palladium (II) complexes and antimicrobial activities - ScienceDirect
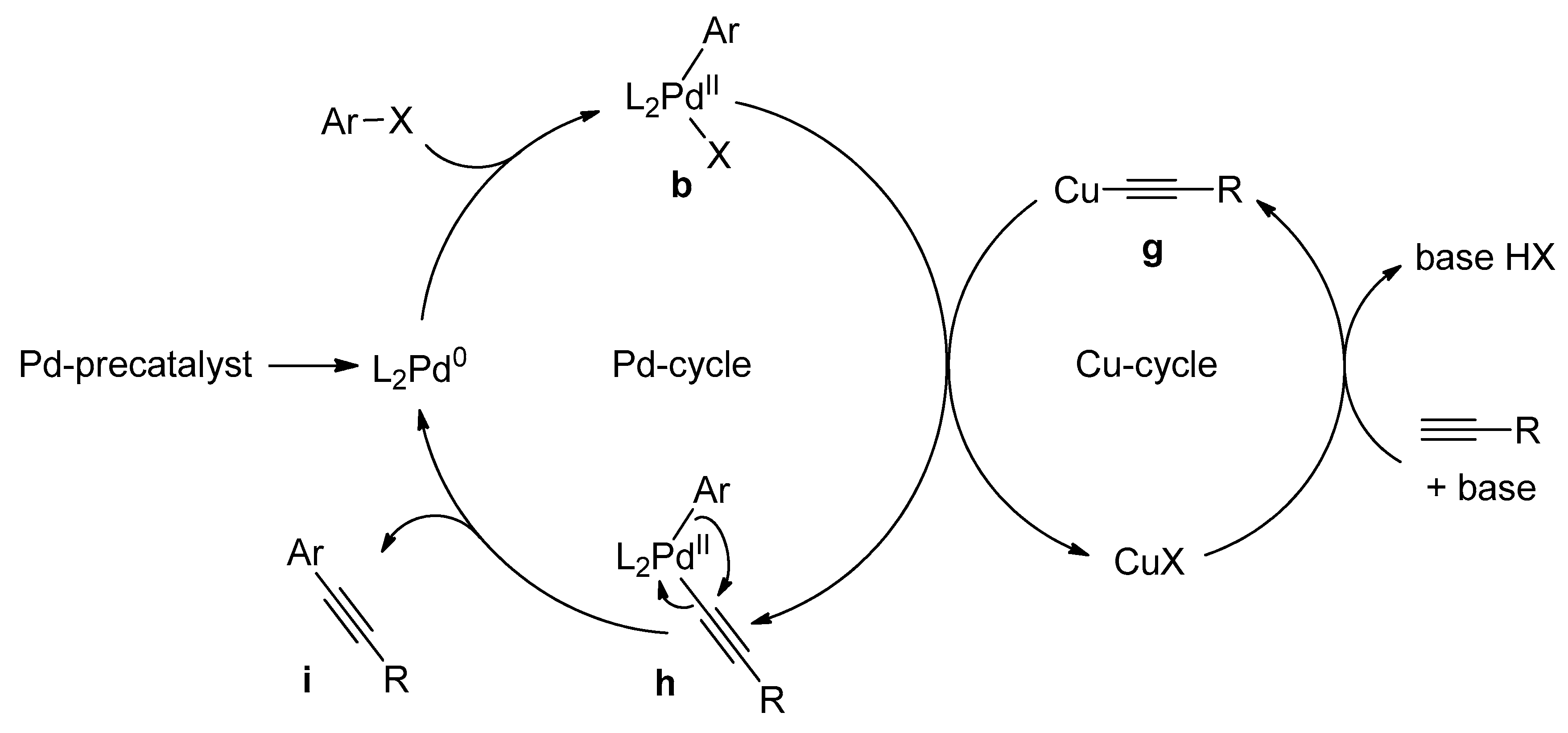
Catalysts | Free Full-Text | Microwave-Assisted Palladium-Catalyzed Cross-Coupling Reactions: Generation of Carbon–Carbon Bond | HTML

Arylation of heterocyclic compounds by benzimidazole-based N-heterocyclic carbene-palladium(II) complexes - ScienceDirect

PDF) Heck reaction using palladium-benzimidazole catalyst: synthesis, characterisation and catalytic activity

Palladium‐ and Copper‐Mediated Direct C‐2 Arylation of Azoles — Including Free (NH)‐Imidazole, ‐Benzimidazole and ‐Indole — Under Base‐Free and Ligandless Conditions - Bellina - 2006 - European Journal of Organic Chemistry - Wiley Online Library



