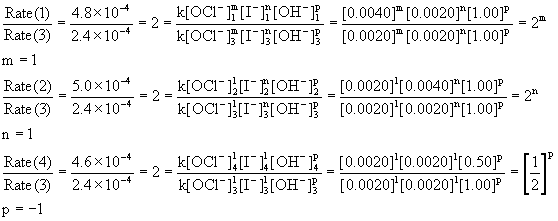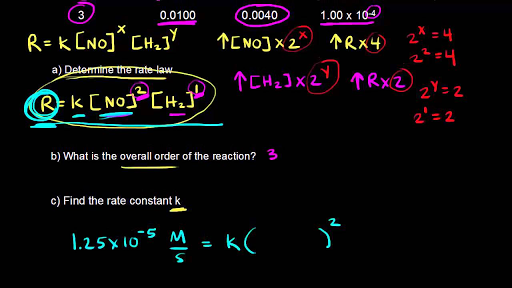Consider the following mechanism:Step 1: 2A ----> B slow.Step 2: B + C ----> D fast - Home Work Help - Learn CBSE Forum

Overall hydrolysis rate constant k hyd for varying pH values at 298 K.... | Download Scientific Diagram

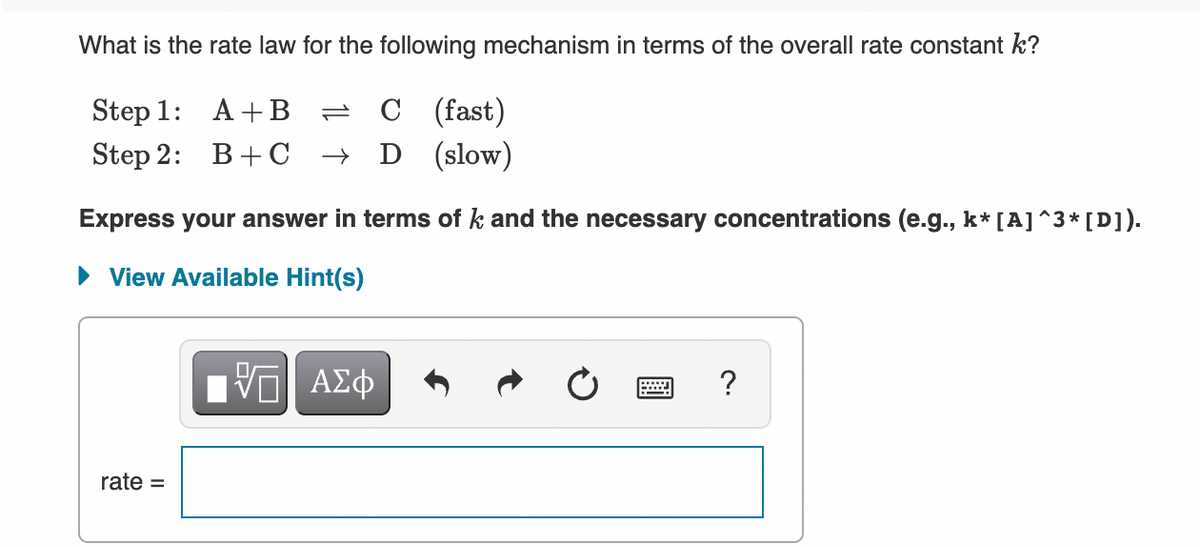
SOLVED: Consider the mechanism Step 1: A B+C Step 2: C+D E Overall: A+D = B+E slow tast Determine the rate law for the overall reaction, where the overall rate constant is

A reaction takes place in three steps. The rate constant are k(1), k(2) and k(3). The overall rate constant k=(k(1)k(3))/(k(2)). If E(1), E(2) and E(3) (energy of activation) are 60, 30 and

OneClass: Consider the following mechanism. step 1: AB+C equilibrium step 2: C+Dâ†' E slow Overall: A...

Determine the rate law for the overall reaction (where the overall rate constant is represented as k)? | Socratic

For a complex reaction A k products Ea1 = 180 kJ/mol ; Ea2 = 80 kJ/mol ; Ea3 = 50 kJ/mol Overall rate constant k is related to individual rate constant by

SOLVED: Consider the mechanism Step I: A = B+C slow Step 2: C+D -E Overall: A+D - B+E Determine the rate law for the overall reaction, where the overall rate constant is

A reaction takes place in three steps. The rate constant of the three steps is K1 , K2 and K3 respectively. The overall rate constant K = K1K3/K2 . The energy of

The Rate Law. Objectives: To understand what a rate law is To determine the overall reaction order from a rate law CLE ppt download