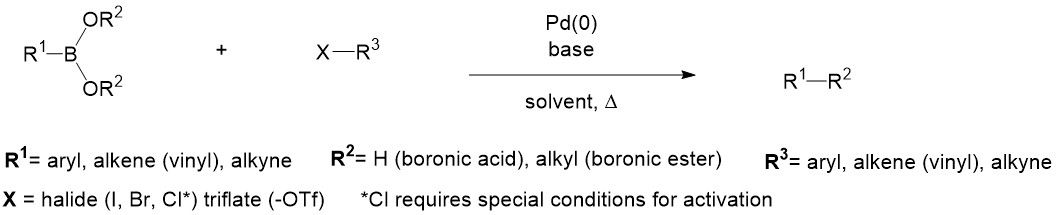
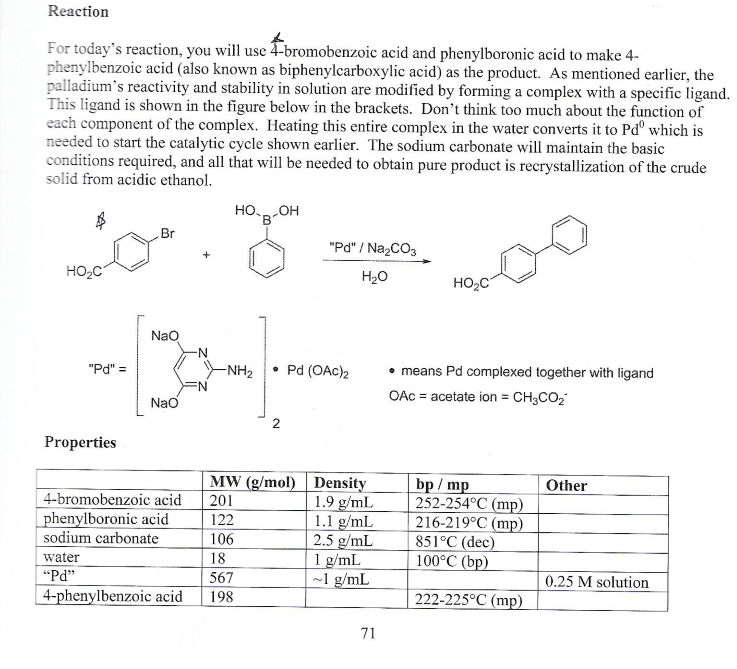
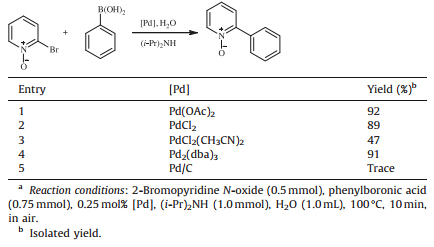
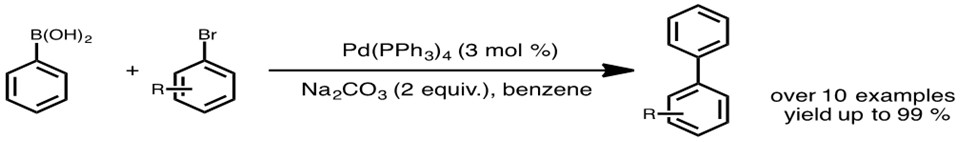PdCl2 Immobilized in Polyacrylamide: a Low Cost and Eco-Friendly Catalyst for Suzuki-Miyaura Reactions
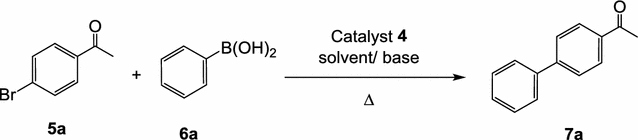
Novel pyridine-based Pd(II)-complex for efficient Suzuki coupling of aryl halides under microwaves irradiation in water | SpringerLink
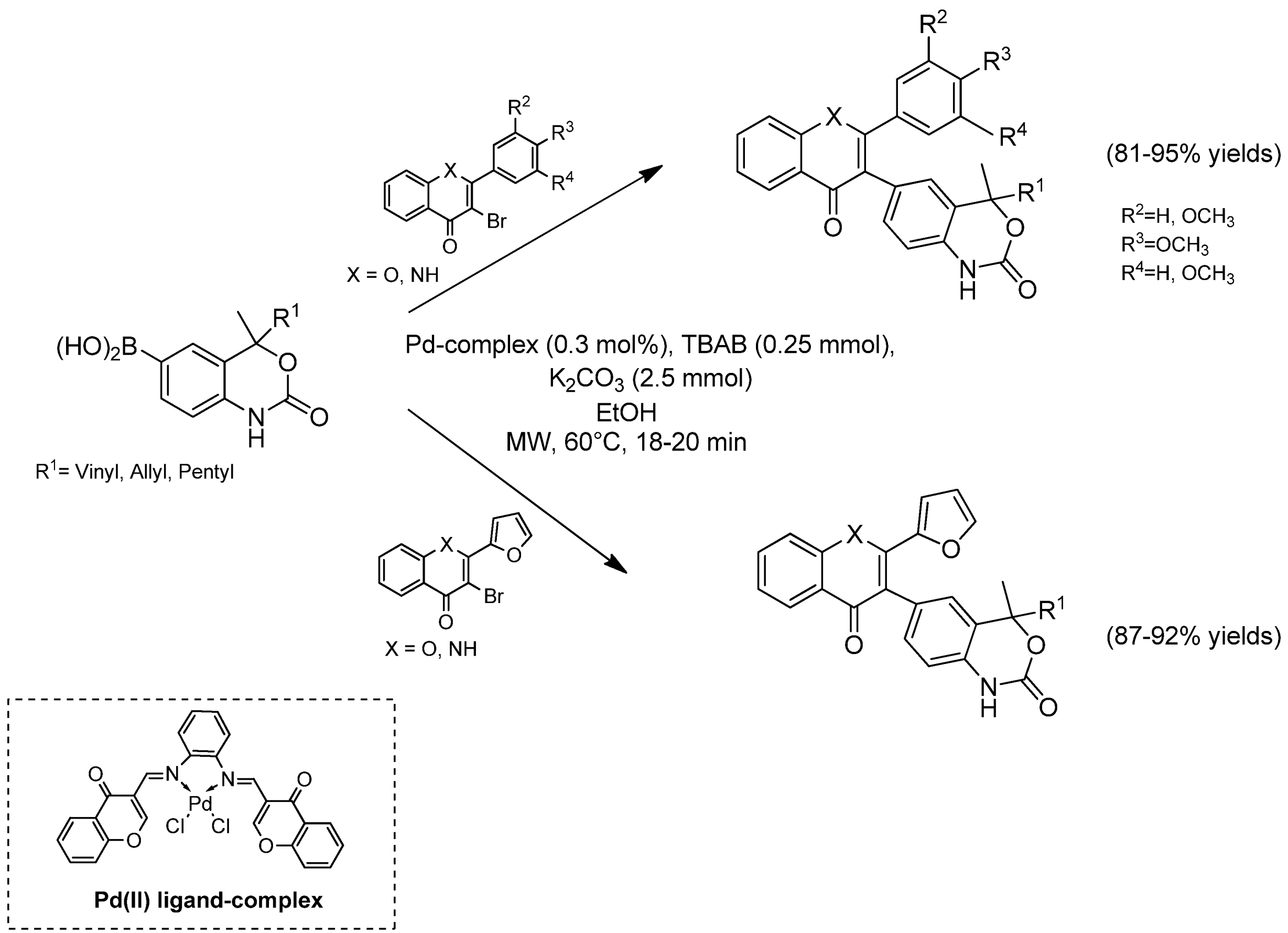
Catalysts | Free Full-Text | Eco-Friendly Physical Activation Methods for Suzuki–Miyaura Reactions | HTML
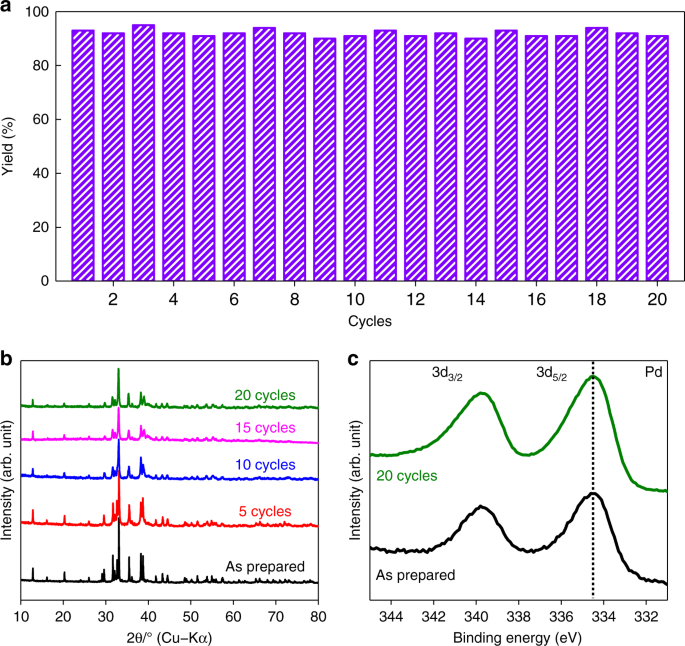
Palladium-bearing intermetallic electride as an efficient and stable catalyst for Suzuki cross-coupling reactions | Nature Communications

Suzuki-Miyaura cross-coupling of phenylboronic acid with aryl halides catalyzed by palladium and nickel species supported on alumina-based oxides - ScienceDirect

Table 6 | Magnetic Mesoporous Silica Nanocomposite Functionalized with Palladium Schiff Base Complex: Synthesis, Characterization, Catalytic Efficacy in the Suzuki–Miyaura Reaction and α-Amylase Immobilization | SpringerLink
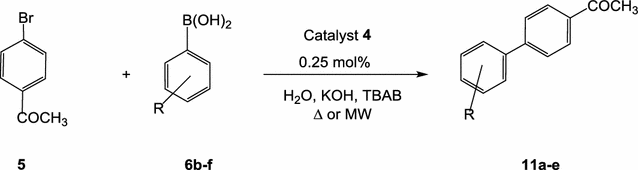
Novel pyridine-based Pd(II)-complex for efficient Suzuki coupling of aryl halides under microwaves irradiation in water | BMC Chemistry | Full Text
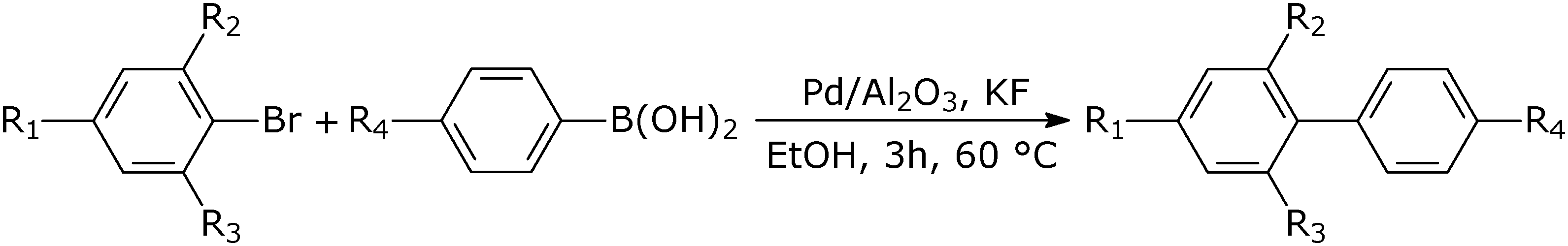
Metallic palladium, PdO, and palladium supported on metal oxides for the Suzuki–Miyaura cross-coupling reaction: a unified view of the process of formation of the catalytically active species in solution - Catalysis Science

Table 1 from New Pd(II) and Pt(II)-diaminophosphine complexes bearing cyclohexyl or isopropyl moiety: use of Pd(II) complexes as precatalyst in Mizoroki{Heck and Suzuki{Miyaura cross-coupling reactions | Semantic Scholar
PLOS ONE: Supported Palladium Nanoparticles Synthesized by Living Plants as a Catalyst for Suzuki-Miyaura Reactions

Catalysts | Free Full-Text | Recent Developments in the Immobilization of Palladium Complexes on Renewable Polysaccharides for Suzuki–Miyaura Cross-Coupling of Halobenzenes and Phenylboronic Acids | HTML

Study of Pd-based catalysts within red algae-derived polysaccharide supports in a Suzuki cross-coupling reaction - RSC Advances (RSC Publishing) DOI:10.1039/C8RA08408D
![Dansyl-PEG-phenylboronic acid | Protein labeling reagent | CAS [N.A.] | Axon 2257 | Axon Ligand™ with >98% purity available from stock from supplier Axon Medchem Dansyl-PEG-phenylboronic acid | Protein labeling reagent | CAS [N.A.] | Axon 2257 | Axon Ligand™ with >98% purity available from stock from supplier Axon Medchem](https://www.axonmedchem.com/media/catalog/product/cache/1/image/9df78eab33525d08d6e5fb8d27136e95/2/2/2257.gif)